Discussion
photons are absorbed by electrons in molecules - the energy is used to excite the electrons, which then slowly return to their ground-state energy... so any coloured substances (e.g. red paint) will absorb some photons in the visible region. obviously colourless stuff like salt or sugar don't.
reflection happens at the interface between two media whenever a wave (light, sound, water) crosses from one medium into another - e.g. from water into concrete, or from air into glass, or from air to metal. basically the incoming wave induces vibrations in the second medium which then re-radiate the wave (i.e. reflect it). the vibrations can be physical (eg water wave, sound) or, when we are talking about light, are vibrations in the polarisation of atoms/molecules (i.e. the distribution of electrons).
reflection happens at the interface between two media whenever a wave (light, sound, water) crosses from one medium into another - e.g. from water into concrete, or from air into glass, or from air to metal. basically the incoming wave induces vibrations in the second medium which then re-radiate the wave (i.e. reflect it). the vibrations can be physical (eg water wave, sound) or, when we are talking about light, are vibrations in the polarisation of atoms/molecules (i.e. the distribution of electrons).
This is one of those simple questions that gets complicated fast and simplified explanations can be a bit misleading (i.e. wrong). So, disclaimer over...
Solids have a bunch of atoms loosely connected together with continuous ranges of allowed vibration energies. If the incoming photon of light has an energy which falls within these ranges then it is absorbed and converted to heat (i.e. vibration). If it doesn't fall within these ranges then it's absorbed and re-emitted almost immediately. In glass the the frequencies of visible light (i.e. the energy of the photons) don't fall in the absorption range unlike, for example, the frequencies of IR photons.
Solids have a bunch of atoms loosely connected together with continuous ranges of allowed vibration energies. If the incoming photon of light has an energy which falls within these ranges then it is absorbed and converted to heat (i.e. vibration). If it doesn't fall within these ranges then it's absorbed and re-emitted almost immediately. In glass the the frequencies of visible light (i.e. the energy of the photons) don't fall in the absorption range unlike, for example, the frequencies of IR photons.
Oxford scientists have created a transparent form of aluminium by bombarding the metal with the world’s most powerful soft X-ray laser. 'Transparent aluminium' previously only existed in science fiction, featuring in the movie Star Trek IV, but the real material is an exotic new state of matter with implications for planetary science and nuclear fusion.
International team, led by Oxford University scientists, report that a short pulse from the FLASH laser ‘knocked out’ a core electron from every aluminium atom in a sample without disrupting the metal’s crystalline structure. This turned the aluminium nearly invisible to extreme ultraviolet radiation. Want to cavet this as the spot with a diameter less than a twentieth of the width of a human hair. At such high intensities the aluminium turned transparent.
International team, led by Oxford University scientists, report that a short pulse from the FLASH laser ‘knocked out’ a core electron from every aluminium atom in a sample without disrupting the metal’s crystalline structure. This turned the aluminium nearly invisible to extreme ultraviolet radiation. Want to cavet this as the spot with a diameter less than a twentieth of the width of a human hair. At such high intensities the aluminium turned transparent.
sort of related... but its a nice video explaining (trying) how light behaves in glass as a wave and as a photon.
http://www.youtube.com/watch?v=CiHN0ZWE5bk
http://www.youtube.com/watch?v=CiHN0ZWE5bk
Otispunkmeyer said:
sort of related... but its a nice video explaining (trying) how light behaves in glass as a wave and as a photon.
http://www.youtube.com/watch?v=CiHN0ZWE5bk
That's teh one I was going to post.http://www.youtube.com/watch?v=CiHN0ZWE5bk
RealSquirrels said:
glass absorbs UV light and infra-red light which is why you won't get sunburnt, but will get hot, sitting inside your greenhouse.
Not entirely true. Glass blocks UVb but not UVa which is how things like this happen.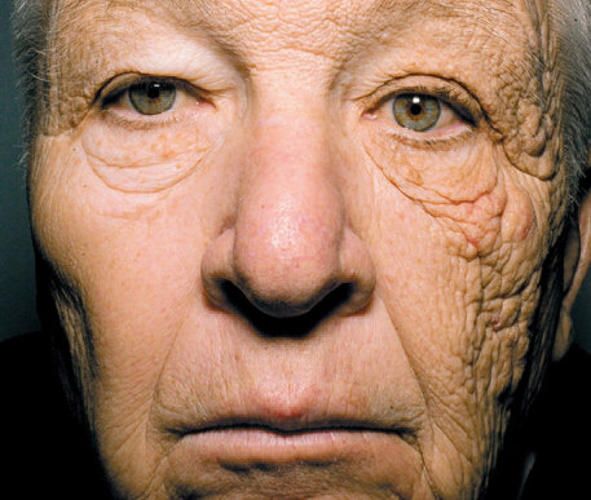
It's also the UVa that causes window tin film to turn purple and eventually clear and why you should always check if a tint job is done with a film that has a UV filter in built. Most do have now.
As I recall from my science workings, on a molecular scale the arrangement of the molecules compared to the wavelength of the radiation (light...) which can pass through. Specifically in the eye, the white of the eye and the cornea are basically made of the same stuff (collagen fibres in an aqueous matrix - the same as cartilage and other connective tissues for those that are interested). But the cornea is transparent because the fibres are in an orderly arrangement with a a specific separation; the white is full of disordered fibres in all directions and of various sizes.
Or magic.
Or magic.
RealSquirrels said:
annodomini2 said:
budfox said:
Well they did transparent aluminium in Star Trek IV
Also known as Sapphire.
There's also Aluminium oxynitride.
Gassing Station | Science! | Top of Page | What's New | My Stuff